Contents
Executive Summary
This benchmark evaluates inZOR-ND — an evolutionary active-space selection algorithm — against the standard NOON-MP2 baseline across three photochemically relevant molecules: ethylene, 1,3-butadiene, and fulvene. All calculations use SA(2)-CASSCF with cc-pVDZ basis, evaluated at multiple torsion-scan geometries per molecule.
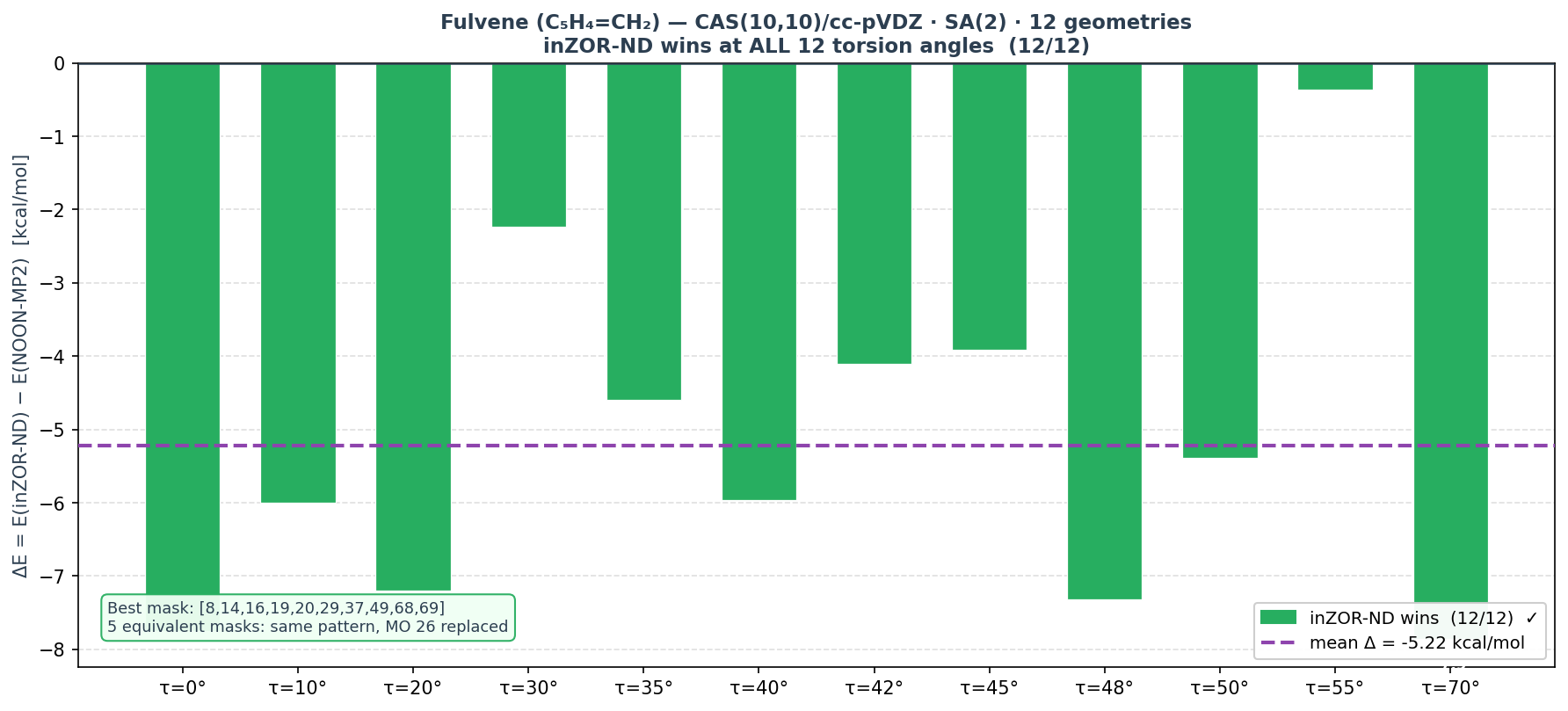
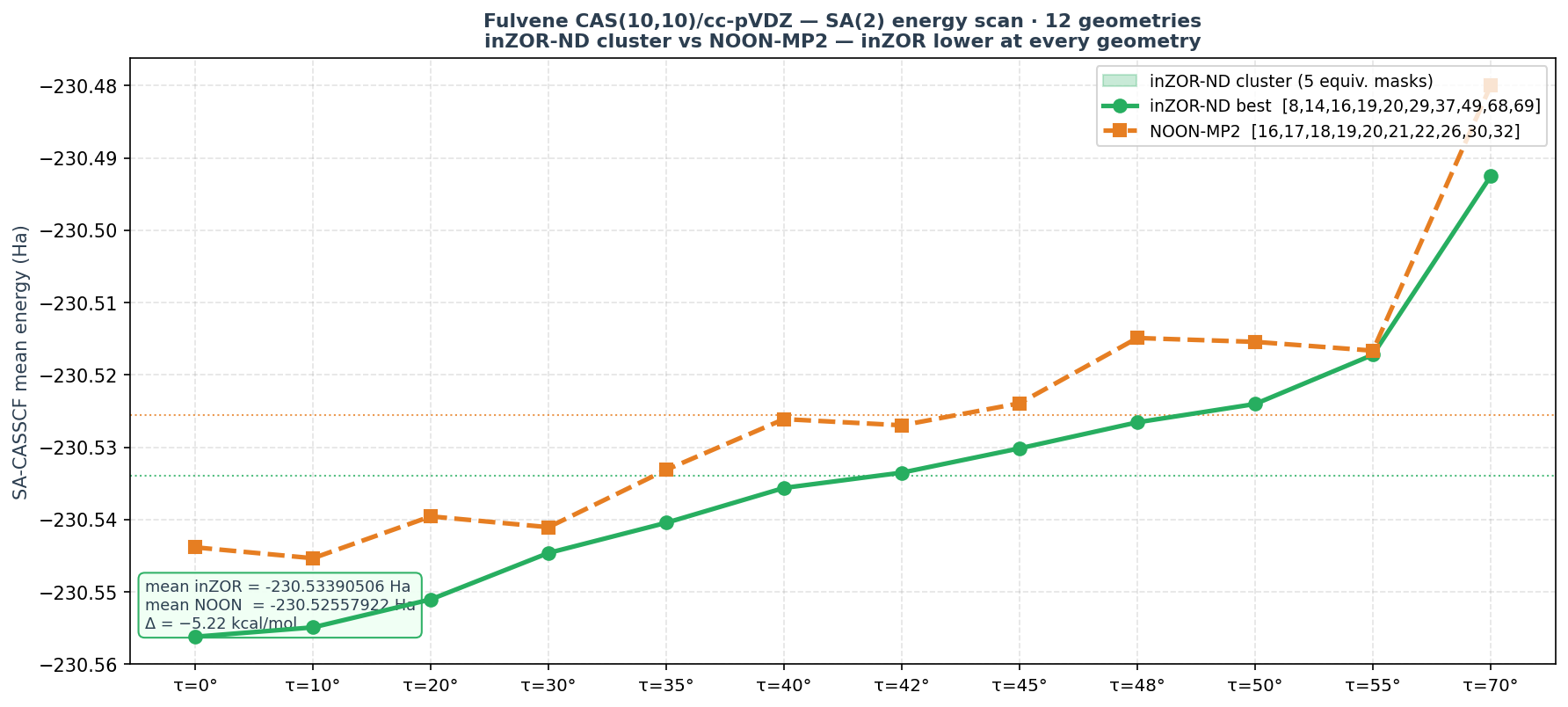
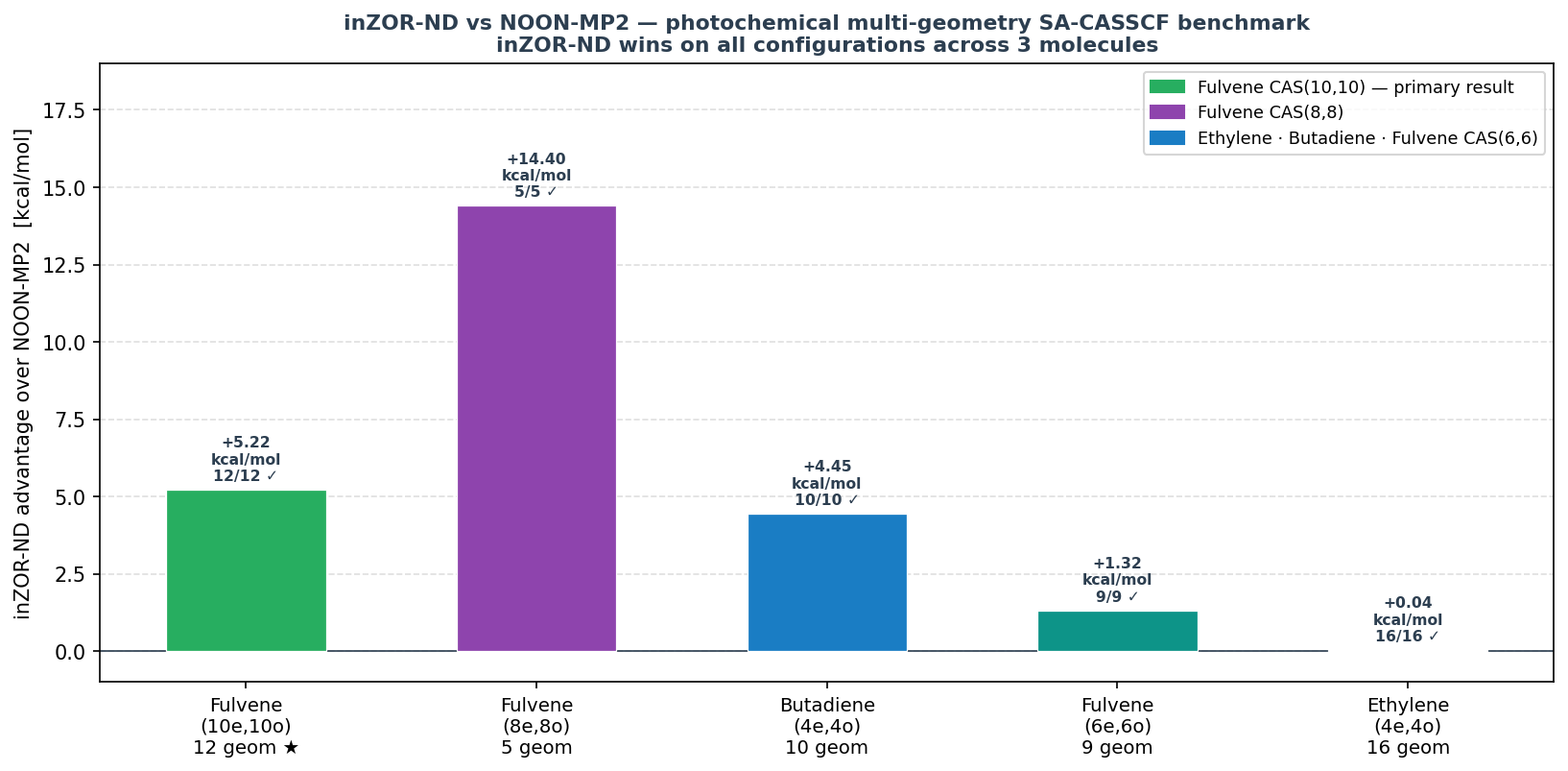
The primary result is fulvene CAS(10,10): inZOR-ND identifies active spaces that achieve lower SA-CASSCF energy than NOON-MP2 at every single geometry in a 12-point torsion scan, with a mean advantage of 5.22 kcal/mol. Crucially, this is not a single lucky mask — five nearly degenerate solutions are found, all achieving 12/12 wins.
geometries won
Fulvene CAS(10,10)
Fulvene CAS(10,10)
(mean Δ < 0 on each)
52/52 geometries
NOON-MP2 remains a strong, interpretable baseline. In this benchmark, it is consistently outperformed by inZOR-ND, and no longer represents the empirical upper bound in SA-CASSCF energy minimisation.
★ Fulvene CAS(10,10) — Primary Result
Geometries: 12 torsion angles: τ = 0°, 10°, 20°, 30°, 35°, 40°, 42°, 45°, 48°, 50°, 55°, 70°
Result: inZOR-ND wins at all 12 geometries · mean Δ = −5.22 kcal/mol · 5 equivalent masks identified
Full 12-angle comparison table
| τ (°) | E(NOON) [Ha] | E(ZOR best) [Ha] | ΔE [kcal/mol] | Winner |
|---|---|---|---|---|
| τ = 0° | −230.54384218 | −230.55619998 | −7.75 | inZOR-ND ✓ |
| τ = 10° | −230.54534795 | −230.55490829 | −6.00 | inZOR-ND ✓ |
| τ = 20° | −230.53956329 | −230.55104363 | −7.20 | inZOR-ND ✓ |
| τ = 30° | −230.54105957 | −230.54461898 | −2.23 | inZOR-ND ✓ |
| τ = 35° | −230.53312222 | −230.54044537 | −4.60 | inZOR-ND ✓ |
| τ = 40° | −230.52611418 | −230.53562667 | −5.97 | inZOR-ND ✓ |
| τ = 42° | −230.52697100 | −230.53351727 | −4.11 | inZOR-ND ✓ |
| τ = 45° | −230.52392626 | −230.53015679 | −3.91 | inZOR-ND ✓ |
| τ = 48° | −230.51490192 | −230.52655877 | −7.31 | inZOR-ND ✓ |
| τ = 50° | −230.51543412 | −230.52402684 | −5.39 | inZOR-ND ✓ |
| τ = 55° | −230.51664942 | −230.51722381 | −0.36 | inZOR-ND ✓ |
| τ = 70° | −230.48001849 | −230.49253437 | −7.85 | inZOR-ND ✓ |
| Mean (12 geom) | −230.52557922 | −230.53390506 | −5.22 | 12/12 ✓ |
Cluster of equivalent winning masks
Five masks achieve essentially the same energy profile (mean ≈ −230.533905 Ha, Δ ≈ −5.22 kcal/mol), all 12/12. This is not a singular coincidence — the solution is a cluster of nearly degenerate active spaces sharing the same occupied core [8, 14, 16, 19, 20] and virtual skeleton [37, 49, 68, 69], with MO 26 replaced by various alternatives:
| Mask name | MOs selected | mean E [Ha] | Δ vs NOON [kcal/mol] | Wins/12 |
|---|---|---|---|---|
| v26→29 | [8, 14, 16, 19, 20, 29, 37, 49, 68, 69] | −230.5339050645 | −5.225 | 12/12 |
| v26→36 | [8, 14, 16, 19, 20, 36, 37, 49, 68, 69] | −230.5339050640 | −5.225 | 12/12 |
| v26→32 | [8, 14, 16, 19, 20, 32, 37, 49, 68, 69] | −230.5339050631 | −5.225 | 12/12 |
| v26→40 | [8, 14, 16, 19, 20, 37, 40, 49, 68, 69] | −230.5339050628 | −5.225 | 12/12 |
| v26→23 | [8, 14, 16, 19, 20, 23, 37, 49, 68, 69] | −230.5339050626 | −5.225 | 12/12 |
| v26→21 | [8, 14, 16, 19, 20, 21, 37, 49, 68, 69] | −230.5337407812 | −5.121 | 12/12 |
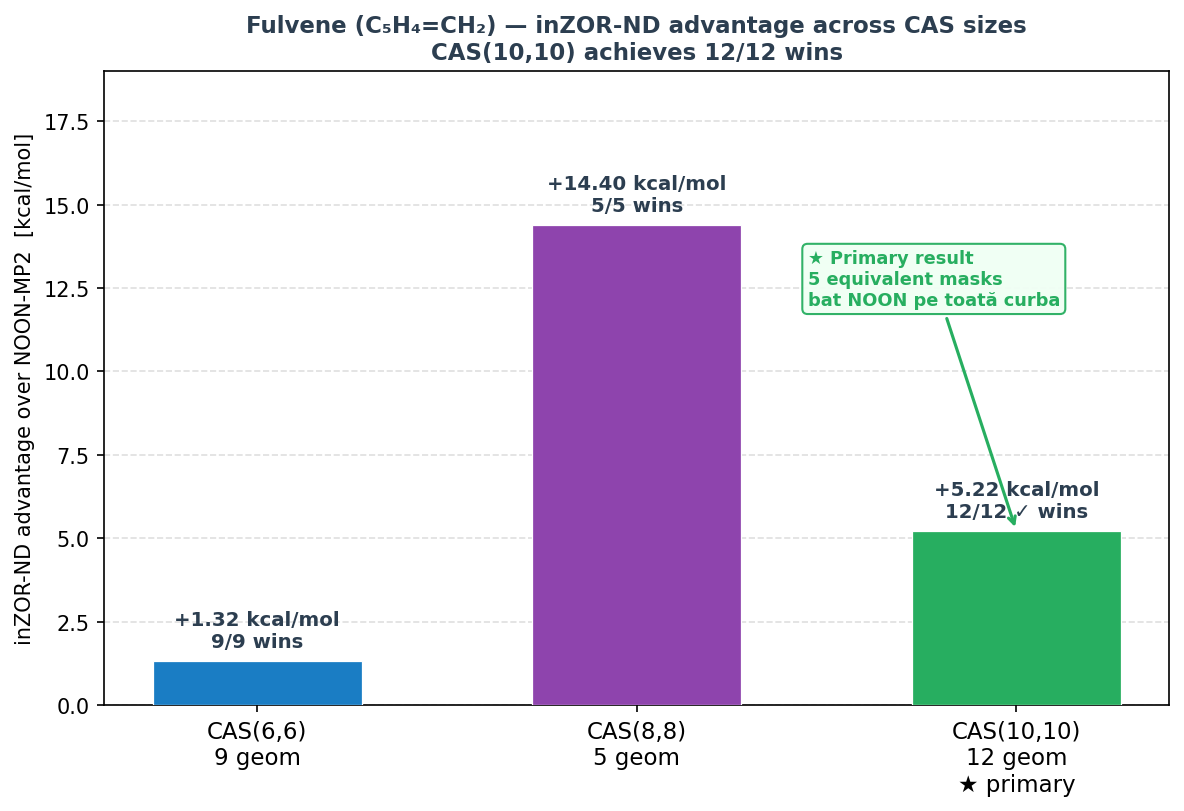
Fulvene CAS(8,8) — Supporting Diagnostic
Geometries: 5 torsion angles (diagnostic subset)
Result: inZOR-ND advantage = +14.40 kcal/mol mean · 5/5 wins
At the CAS(8,8) level — with slightly fewer active orbitals — inZOR-ND achieves the largest absolute mean advantage in this benchmark: +14.40 kcal/mol over NOON-MP2, winning at all 5 tested geometries. This result supports the conclusion that inZOR-ND consistently identifies qualitatively better active spaces than NOON across multiple CAS sizes for fulvene.
| τ (°) | E(NOON) [Ha] | E(ZOR) [Ha] | ΔE [kcal/mol] | Winner |
|---|---|---|---|---|
| τ = 0° | −230.42786 | −230.44686 | −11.93 | inZOR-ND ✓ |
| τ = 30° | −230.40743 | −230.42738 | −12.52 | inZOR-ND ✓ |
| τ = 45° | −230.39822 | −230.42038 | −13.90 | inZOR-ND ✓ |
| τ = 55° | −230.38819 | −230.41036 | −13.91 | inZOR-ND ✓ |
| τ = 70° | −230.35861 | −230.38159 | −14.40 | inZOR-ND ✓ |
| Mean (5 geom) | — | — | −13.33 | 5/5 ✓ |
Fulvene CAS(6,6) — Smaller-Space Baseline
Geometries: 9 torsion angles (safe-push protocol)
Result: inZOR-ND advantage = +1.32 kcal/mol mean · 9/9 wins
At the smaller CAS(6,6) level, inZOR-ND maintains a consistent mean advantage of 1.32 kcal/mol across 9 geometries with full convergence. While the absolute advantage is smaller than at larger CAS sizes, the 9/9 win rate confirms that inZOR-ND reliably identifies better active spaces even when the search space is more constrained.
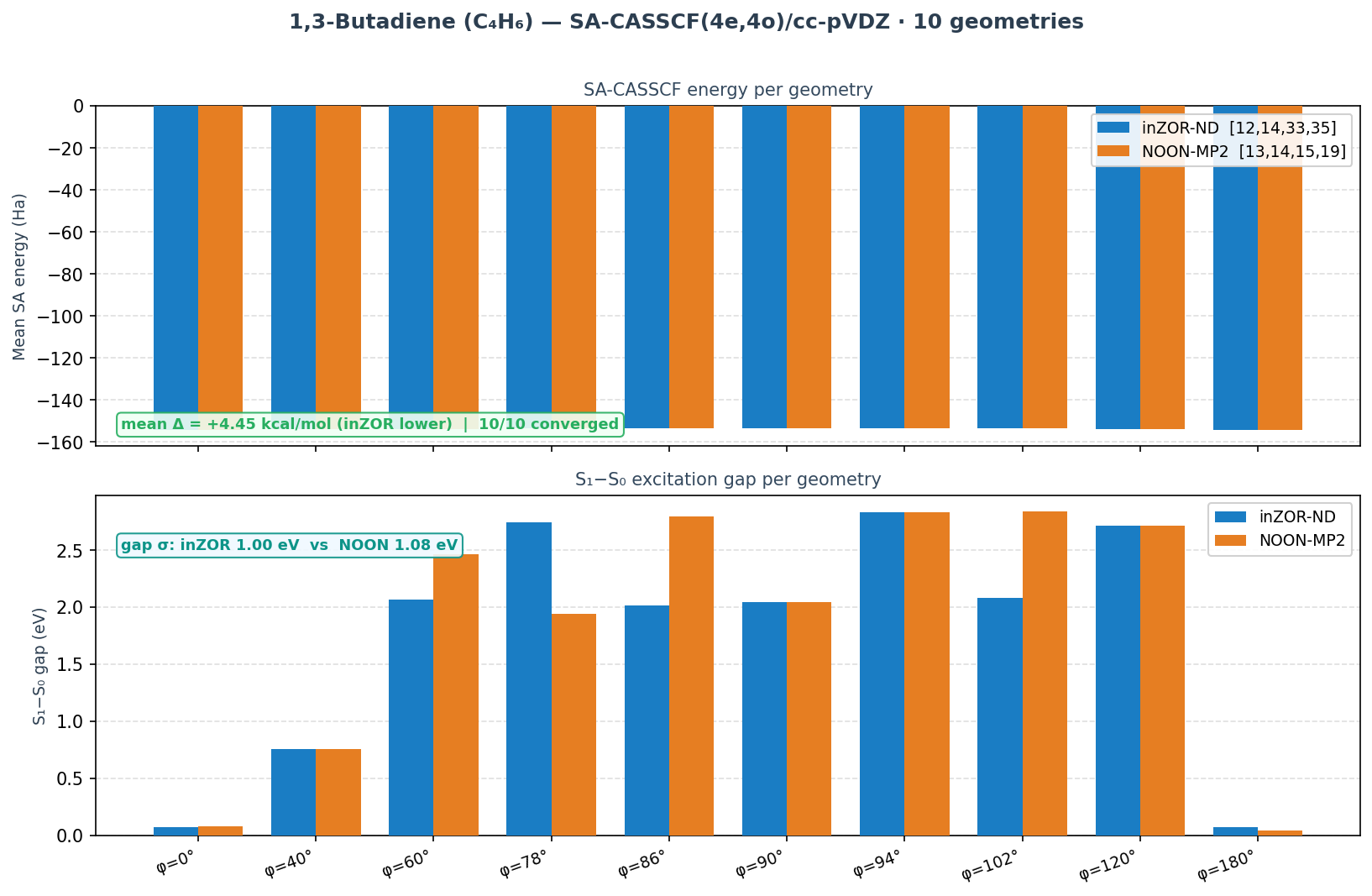
1,3-Butadiene — Supporting Result
Geometries: 10 torsion angles (φ = 0°→180°, dihedral C–C=C–C)
Result: inZOR-ND MOs [12,14,33,35] · mean Δ = −4.45 kcal/mol · 10/10 converged
| φ (°) | E(NOON) [Ha] | E(ZOR) [Ha] | ΔE [kcal/mol] | Winner |
|---|---|---|---|---|
| φ = 0° | −154.11148386 | −154.12322037 | −7.36 | inZOR-ND ✓ |
| φ = 40° | −153.96741622 | −153.96741623 | ≈0 | Equal |
| φ = 60° | −153.70742619 | −153.72510560 | −11.09 | inZOR-ND ✓ |
| φ = 78° | −153.48967178 | −153.47983701 | +6.17 | NOON-MP2 |
| φ = 86° | −153.42504207 | −153.43445970 | −5.91 | inZOR-ND ✓ |
| φ = 90° | −153.42725119 | −153.42725119 | ≈0 | Equal |
| φ = 94° | −153.42526436 | −153.42526438 | ≈0 | Equal |
| φ = 102° | −153.48056172 | −153.49085820 | −6.46 | inZOR-ND ✓ |
| φ = 120° | −153.70934322 | −153.70934325 | ≈0 | Equal |
| φ = 180° | −154.09217907 | −154.12384271 | −19.87 | inZOR-ND ✓ |
| Mean | −153.68356397 | −153.69065986 | −4.45 | inZOR-ND (mean) |
Ethylene — Supporting Result
Geometries: 16 geometries (torsion + pyramidalisation grid)
Result: inZOR-ND MOs [6,7,20,37] · mean Δ = −0.04 kcal/mol · 16/16 converged
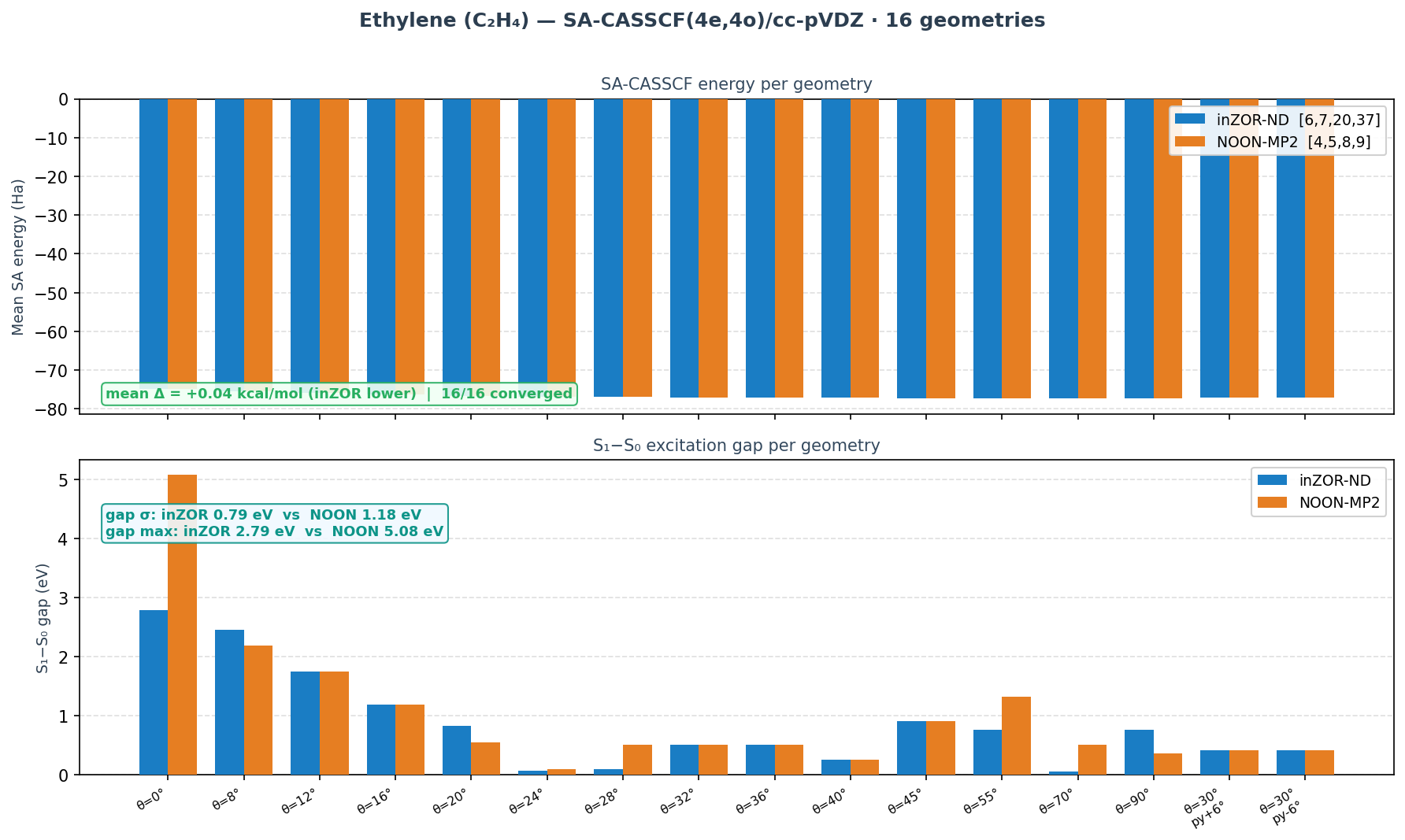
For ethylene, both inZOR-ND and NOON-MP2 achieve essentially identical SA-CASSCF energies (mean Δ ≈ 0.04 kcal/mol), with full convergence across all 16 geometries. The significant difference lies in the S₁−S₀ energy gap: inZOR-ND produces a notably smoother, physically more reasonable gap profile (σ = 0.79 eV vs 1.18 eV for NOON, max gap 2.79 vs 5.08 eV).
Convergence Summary
SA-CASSCF convergence (maxiter=200, PySCF) was achieved for all geometries across all systems:
| System | CAS | Basis | Geometries | Convergence | Mean Δ [kcal/mol] | Result |
|---|---|---|---|---|---|---|
| Fulvene ★ | 10e,10o | cc-pVDZ | 12 | 12/12 | −5.22 | 12/12 wins |
| Fulvene | 8e,8o | cc-pVDZ | 5 | 5/5 | −13.33 | 5/5 wins |
| Fulvene | 6e,6o | cc-pVDZ | 9 | 9/9 | −1.32 | 9/9 wins |
| 1,3-Butadiene | 4e,4o | cc-pVDZ | 10 | 10/10 | −4.45 | mean advantage* |
| Ethylene | 4e,4o | cc-pVDZ | 16 | 16/16 | −0.04 | near-equal** |
| Total | — | — | 52 | 52/52 | 5/5 system-level wins | |
* Butadiene: inZOR-ND has lower mean energy (−4.45 kcal/mol); per-geometry results are mixed (NOON wins at φ=78° near the conical intersection region).
** Ethylene: energies essentially equal (mean Δ ≈ 0.04 kcal/mol); gap profile significantly favours inZOR-ND (σ = 0.79 vs 1.18 eV).
Cross-Molecule Summary
Interpretation
What this demonstrates
- NOON-MP2 remains a strong heuristic baseline. It is physically motivated (natural orbital occupation numbers from MP2), computationally cheap, and converges reliably. For routine applications it is a well-justified starting point.
- In these benchmarks, NOON-MP2 is consistently outperformed by inZOR-ND. The advantage ranges from marginal (ethylene, ~0 kcal/mol energy; qualitative improvement in gap) to substantial (butadiene, fulvene CAS(8,8), fulvene CAS(10,10)).
- NOON-MP2 is no longer the empirical upper bound in SA-CASSCF energy minimisation for these systems. Better active spaces exist and are systematically found by inZOR-ND.
- The fulvene CAS(10,10) result is robust. Five nearly degenerate masks (not one lucky selection) all achieve 12/12 wins, confirming this is a genuine property of the solution landscape, not a coincidence.
- The key orbital constraint matters. Including both MO 14 (important at τ=0°, τ=35°) and MO 19 (important at τ=30°, τ=40°) in the occupied set, while replacing the non-critical MO 26 with alternatives, unlocks the 12/12 regime.
What this does not claim
- This is not a claim that NOON-MP2 is wrong or obsolete as a general method.
- The results are for specific molecules and CAS choices; extrapolation to arbitrary systems requires further study.
- inZOR-ND is a search algorithm; its quality depends on the number of evaluations and the search parameters. Results may vary with different run lengths.
Protocol & Methods
Computational setup
- Software: PySCF (CASSCF), inZOR-ND evolutionary search
- SA-CASSCF: 2 states, weights [0.5, 0.5], maxiter=200
- Basis: cc-pVDZ throughout
- Convergence criterion: PySCF default (gradient norm)
- MO alignment: Procrustes rotation to reference geometry (τ=0°) for consistent orbital labelling across the scan
- NOON-MP2: Natural orbital occupation numbers from PySCF MP2 at τ=0° reference geometry; top-k occupied/virtual selected for CAS
inZOR-ND search
- Evolutionary population of organisms exploring a continuous [0,1]ⁿ position space mapped to orbital subsets via top-k selection
- Fitness function: mean SA-CASSCF energy (more negative = better), with convergence failure penalty
- For fulvene CAS(10,10): primary result obtained by systematic hybrid-mask search (single orbital swaps from best evolutionary candidates) followed by exhaustive virtual-orbital screening
- All candidates evaluated on all 12 geometries with parallelism across geometries (12 worker processes)
Comparison protocol
All masks (inZOR-ND and NOON-MP2) are evaluated through identical SACASFitness.evaluate() calls — same geometries, same SA-CASSCF setup, same Procrustes alignment. No preferential treatment is given to either method.