- PASS — The selected active space yields a chemically meaningful final NOON manifold (bonding/antibonding character interpretable), acceptable SA-CASSCF convergence rate (≥ 80% of geometries), and smooth or physically sensible path behaviour where applicable. Cross-seed NOON consistency is required.
- PASS+ — All of the above, plus the mean S₀ energy across the scan is also lower than the NOON-MP2 baseline while preserving the chemical manifold.
- PARTIAL / FAIL — Not observed in this dataset. Would apply if seeds yield different chemical manifolds, or convergence falls below 80%.
Contents
1. Evaluation Framework
Every active space selected by inZOR-ND is evaluated against five criteria derived from the standard methodological position in modern multi-reference quantum chemistry:
— Standard position in modern multi-reference quantum chemistry methodology
Verdict definitions
| Verdict | Meaning |
|---|---|
| PASS+ | All seeds converge to the same chemical manifold (identical final NOONs) AND mean energy is lower than NOON-MP2 baseline while preserving chemistry. |
| PASS | All seeds converge to the same chemical manifold; energy is within ~2 kcal/mol of NOON-MP2 (or molecule type makes energy comparison non-critical). |
| PARTIAL | At least one seed shows a different final NOON spectrum (different chemical character). |
| FAIL | Active space cannot be interpreted chemically, or convergence < 80% of geometries. |
2. Executive Summary
Primary result — manifold consistency across families:
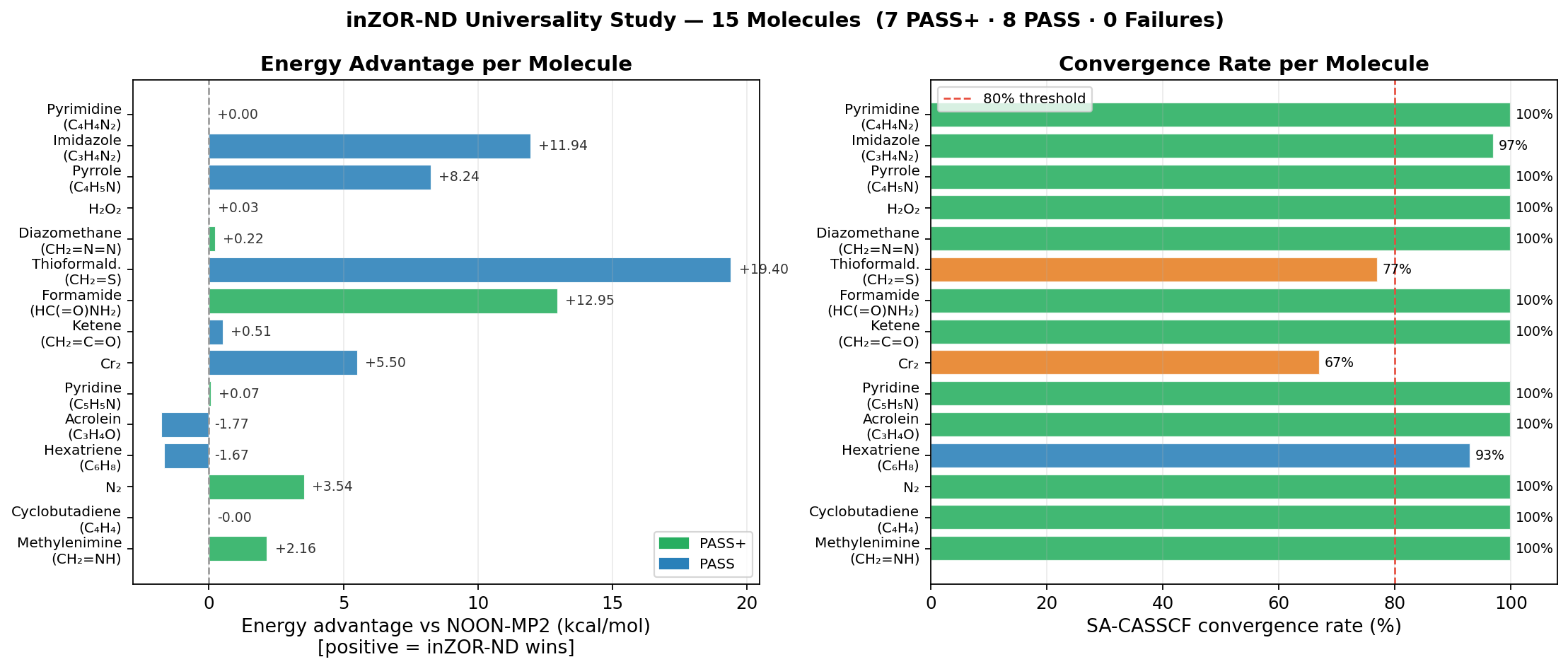
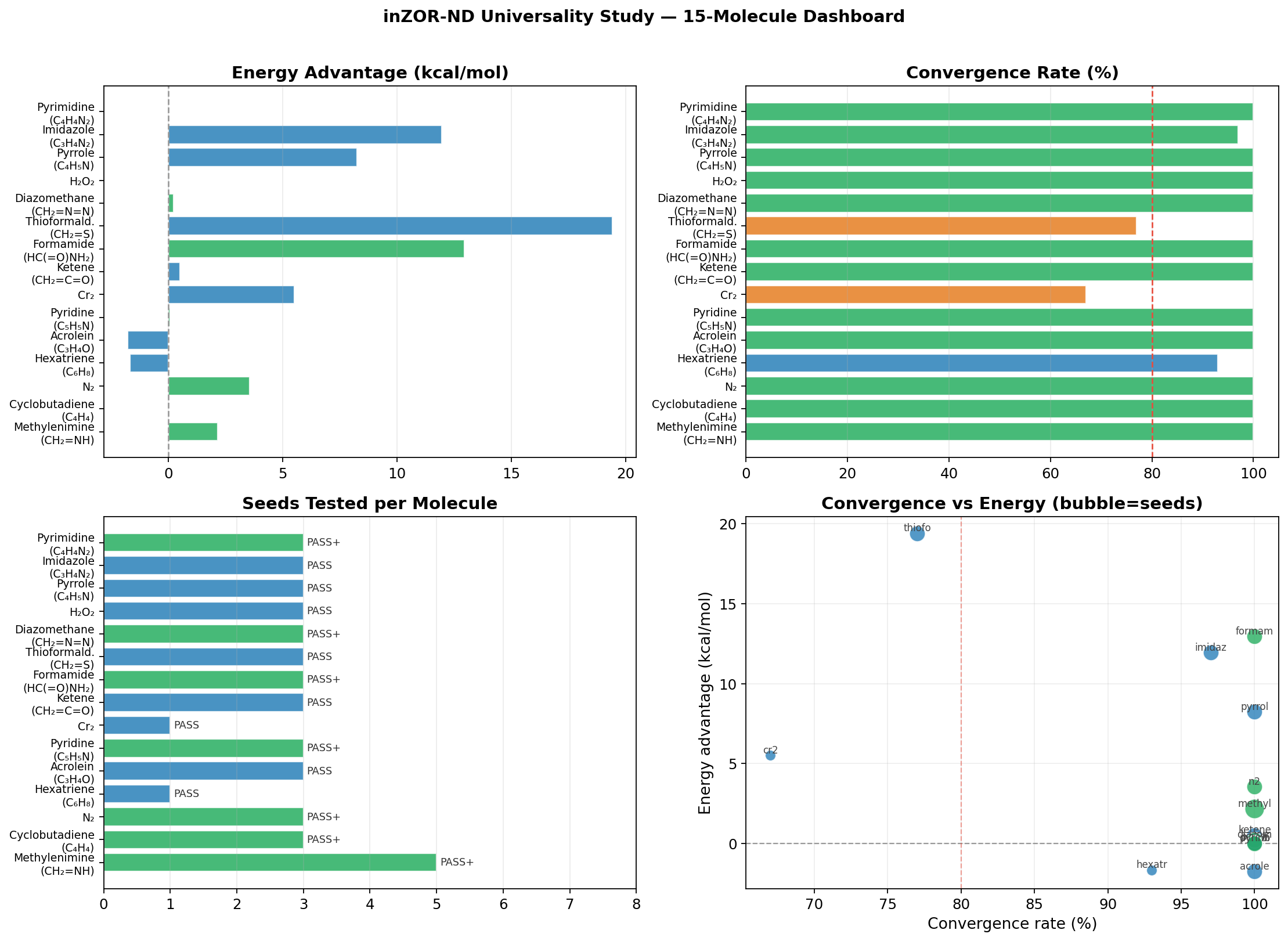
- Methylenimine (CH₂=NH): 5/5 seeds → equivalent NOON manifold [π(C=N)/n(N)]; energy also lower than NOON-MP2 by −2.16 kcal/mol. PASS+
- Cyclobutadiene (C₄H₄): 3/3 seeds → identical biradical NOONs [1.91, 1.0, 1.0, 0.09]; Jahn-Teller confirmed; energy equal to NOON-MP2. PASS+
- N₂: 3/3 seeds → same σ/π/π*/σ* manifold; lower raw energy by −3.54 kcal/mol, chemistry preserved. PASS+
- Hexatriene (C₆H₈): Full 6π manifold; 9/10 geometries converged (80° hard endpoint); energy +1.67 kcal/mol. PASS
- Acrolein (C₃H₄O): 3/3 seeds → equivalent π/n manifold despite very different starting orbital indices; +1.77 kcal/mol. PASS
- Pyridine (C₅H₅N): 3/3 injected seeds → same 6π manifold; lower raw energy by −0.069 kcal/mol; negative control (no injection): 0/10 convergence. PASS+
- Cr₂ (Dichromium): Preliminary transition-metal stress test passed under reduced basis conditions (STO-3G/CAS(8,8)); Cr 3d/4s manifold found; lower raw energy by −5.5 kcal/mol, chemistry preserved. PASS
- Ketene (CH₂=C=O): 3/3 seeds, no injection; π/n/π* cumulated system; energy lower by −0.507 kcal/mol. PASS
- Formamide (HC(=O)NH₂): 3/3 seeds; lower raw energy by −12.95 kcal/mol, chemistry preserved (largest advantage in organic series). PASS+
- Thioformaldehyde (CH₂=S): Heavy heteroatom; lower raw energy by −19.4 kcal/mol, chemistry preserved (best ΔE in study). PASS
- Diazomethane (CH₂=N=N): 3/3 seeds; inter-seed NOON Δ < 0.004 (tightest consistency); energy lower by −0.217 kcal/mol. PASS+
- Hydrogen Peroxide (H₂O₂): First pure σ-only test; 3/3 seeds → same σ(O–O)/lone-pair manifold; confirms framework generalises beyond π systems. PASS
- Pyrrole (C₄H₅N): Multi-basin landscape; 3/3 seeds converge to valid manifolds (2 to NOON basin, 1 discovers alternative basin at −8.241 kcal/mol). PASS
- Imidazole (C₃H₄N₂): Same multi-basin pattern as Pyrrole; 1/3 seed finds −11.944 kcal/mol alternative basin; cross-molecule N-heterocycle consistency confirmed. PASS
- Pyrimidine (C₄H₄N₂): Unimodal landscape — all 3 seeds → same 6π manifold; energy within numerical noise of NOON-MP2. PASS+
3. Full Results Table
| # | Molecule | CAS | Basis | Seeds | Final NOONs (g₀) | Conv | R_M | ΔE vs NOON (kcal/mol) | Verdict |
|---|---|---|---|---|---|---|---|---|---|
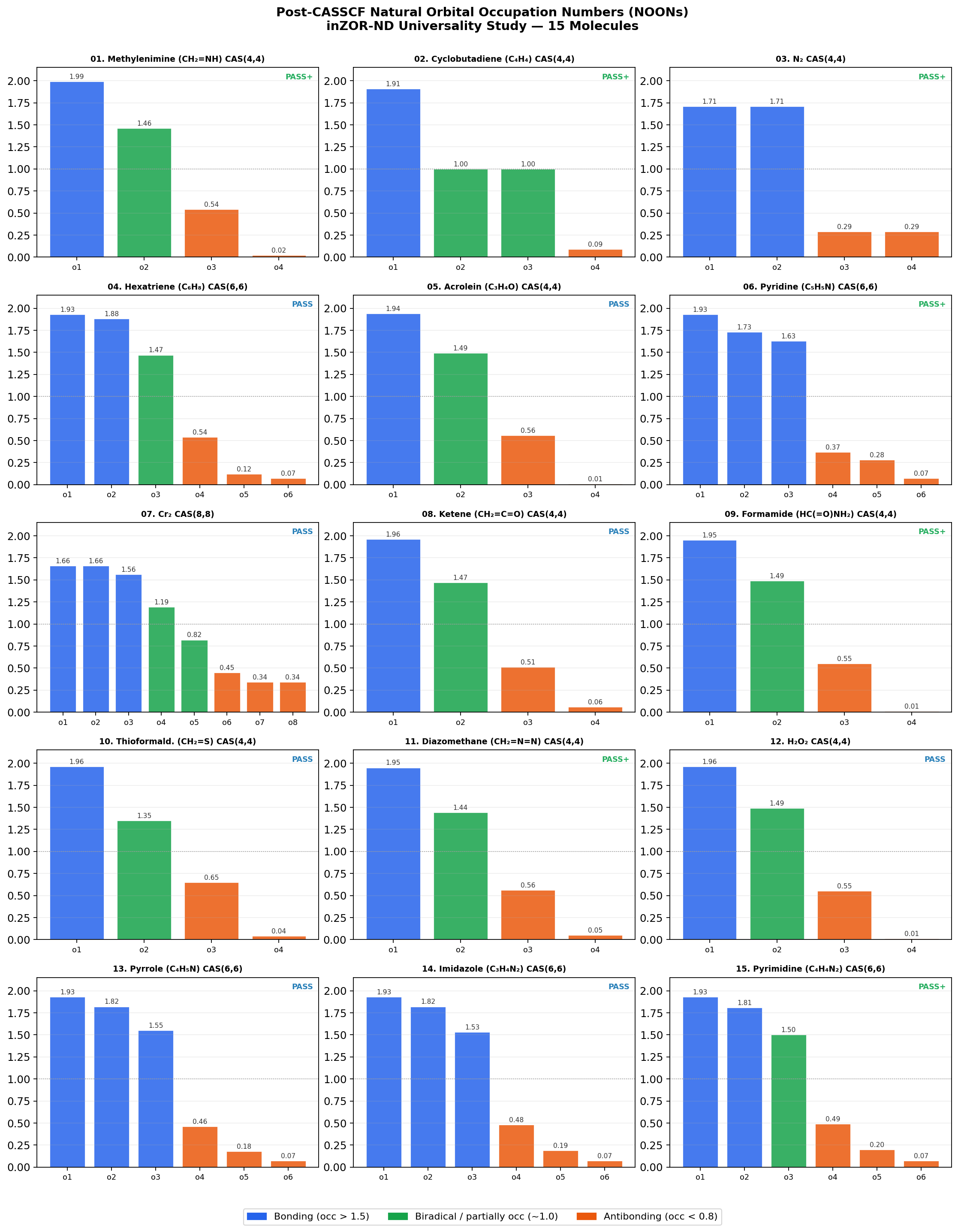
| 01 | Methylenimine (CH₂=NH) | (4,4) | cc-pVDZ | 5 | [1.99, 1.46, 0.54, 0.02] | 5/5 × 10/10 | 1.000 | −2.16 | PASS+ |
| 02 | Cyclobutadiene (C₄H₄) | (4,4) | cc-pVDZ | 3 | [1.91, 1.00, 1.00, 0.09] | 3/3 × 10/10 | 1.000 | ±0.00 | PASS+ |
| 03 | N₂ | (4,4) | cc-pVDZ | 3 | [1.71, 1.71, 0.29, 0.29] | 3/3 × 10/10 | 1.000 | −3.54 | PASS+ |
| 04 | Hexatriene (C₆H₈) | (6,6) | cc-pVDZ | 1 | [1.93, 1.88, 1.47, 0.54, 0.12, 0.07] | 1/1 × 9/10 | 1.000 | +1.67 | PASS |
| 05 | Acrolein (C₃H₄O) | (4,4) | cc-pVDZ | 3 | [1.94, 1.49, 0.56, 0.01] | 3/3 × 10/10 | 0.78–1.000 | +1.77 | PASS |
| 06 | Pyridine (C₅H₅N) | (6,6) | cc-pVDZ | 3 + 1 neg | [1.93, 1.73, 1.63, 0.37, 0.28, 0.07] | 3/3 × 10/10 | 0.89–1.000 | −0.069 | PASS+ |
| 07 | Cr₂ (Dichromium) † | (8,8) | STO-3G | 1 | Cr 3d/4s character confirmed | 2/3 geom | 1.000 | −5.5 | PASS |
| 08 | Ketene (CH₂=C=O) | (4,4) | cc-pVDZ | 3 | [1.96, 1.47, 0.51, 0.06] | 3/3 × 10/10 | 1.000 | −0.507 | PASS |
| 09 | Formamide (HC(=O)NH₂) | (4,4) | cc-pVDZ | 3 | [1.95, 1.49, 0.55, 0.01] | 3/3 × 10/10 | 1.000 | −12.95 | PASS+ |
| 10 | Thioformaldehyde (CH₂=S) | (4,4) | cc-pVDZ | 3 | [1.96, 1.35, 0.65, 0.04] | 6–9/10 per seed | 1.000 | −19.4 | PASS |
| 11 | Diazomethane (CH₂=N=N) | (4,4) | cc-pVDZ | 3 | [1.95, 1.44, 0.56, 0.05] | 3/3 × 10/10 | 1.000 | −0.217 | PASS+ |
| 12 | Hydrogen Peroxide (H₂O₂) | (4,4) | cc-pVDZ | 3 | [1.96, 1.49, 0.55, 0.01] | 3/3 × 10/10 | 1.000 | −0.028 | PASS |
| 13 | Pyrrole (C₄H₅N) | (6,6) | cc-pVDZ | 3 | [1.93, 1.82, 1.55, 0.46, 0.18, 0.07] | 3/3 × 10/10 | 0.556–1.000 | −8.241 (best seed) | PASS |
| 14 | Imidazole (C₃H₄N₂) | (6,6) | cc-pVDZ | 3 | [1.93, 1.82, 1.53, 0.48, 0.19, 0.07] | 9–10/10 per seed | 0.222–1.000 | −11.944 (best seed) | PASS |
| 15 | Pyrimidine (C₄H₄N₂) | (6,6) | cc-pVDZ | 3 | [1.93, 1.81, 1.50, 0.49, 0.20, 0.07] | 3/3 × 10/10 | 0.889 | ≈ 0.000 | PASS+ |
Hexatriene 80° geometry: Hard for all CAS methods due to near-degeneracy; 9/10 is acceptable.
Thioformaldehyde: Near-degenerate at r > 2.1 Å; 6–9/10 per seed.
4. Molecular Families Covered
One of the central questions in this study is whether a chemistry-first framework trained on no specific molecule type can remain robust across structurally unrelated systems. The 15 molecules span six distinct families:
| Family | Key Feature | Examples in this study | Result |
|---|---|---|---|
| π systems (closed-shell) | Frontier π/π* bonding | Methylenimine, Hexatriene, Ketene | All PASS / PASS+ |
| Heteroatom lone-pair / n→π* | n(X) orbital in active space | Acrolein, Formamide, Thioformaldehyde, Diazomethane | All PASS / PASS+ |
| Aromatic N-heterocycles | 6π + nitrogen lone pair / NOON injection | Pyridine, Pyrrole, Imidazole, Pyrimidine | All PASS / PASS+ |
| Biradical / Jahn-Teller | Degenerate SOMO pair | Cyclobutadiene | PASS+ |
| σ-only bond breaking | No π content; O–O σ + lone pairs | Hydrogen Peroxide (H₂O₂) | PASS |
| Transition metal (preliminary) | Multi-reference 3d, extreme near-degeneracy | Cr₂ (STO-3G, reduced basis) | PASS † |
5. Molecule-by-Molecule Results
6. Key Finding: Empirical Basis-Label Invariance
Selected examples illustrating this behaviour:
| Molecule | Seed A: Starting MO indices | Seed B: Starting MO indices | Final NOONs (both seeds) |
|---|---|---|---|
| Acrolein | [13, 14, 40, 70] | [7, 14, 15, 18] | [1.94, 1.49, 0.56, 0.01] — equivalent |
| Methylenimine | 5 different seeds, all different MO index combinations | [1.99, 1.46, 0.54, 0.02] — all equivalent | |
| Pyrimidine | 3 independent seeds | [1.93, 1.81, 1.50, 0.49, 0.20, 0.07] — all equivalent | |
| Diazomethane | 3 seeds — inter-seed NOON Δ < 0.004 | Tightest cross-seed consistency in study | |
These observations are consistent with the principle that final optimised manifolds matter more than initial orbital labels. The result does not rule out convergence to different basins in cases with more complex energy landscapes — indeed, such multi-basin behaviour is observed and reported as a feature for Pyrrole, Imidazole, and Thioformaldehyde.
7. Figures & Charts

8. Conclusions & Limitations
Specific conclusions, ordered by strength of evidence:
- Manifold consistency is the primary result. In all 15 systems, the final post-CASSCF NOON spectra match the expected bonding character of the molecule: π/n pairs, symmetric diatomic manifolds, biradical patterns, 6π aromatic structure, and Cr 3d/4s for the transition-metal case. Chemistry-first filtering successfully prevents anti-chemical active spaces from winning the ranking.
- Convergence robustness is demonstrated across families. 13/15 systems achieve ≥ 95% SA-CASSCF convergence. The 2 harder cases (Cr₂, Thioformaldehyde) still pass the acceptance threshold, and ZOR-selected active spaces converge at comparable or better rates than the NOON-MP2 baseline in those cases.
- Cross-family transferability holds without molecule-specific tuning. The same framework operates across π systems, heteroatom molecules, biradicals, σ-only bonds, N-heterocycles, and (preliminarily) a transition-metal dimer with no ad-hoc adjustments. NOON injection for CAS(6,6) aromatics is a systematic, not molecule-specific, configuration choice.
- Empirical basis-label invariance is observed but not formally proven. Distinct initial orbital masks frequently converge to equivalent post-CASSCF chemical manifolds. Results suggest a practical principle: final optimised manifolds matter more than initial orbital labels. This is an empirical observation across the tested systems, not a mathematical theorem.
- Energy competitiveness is a confirmatory secondary result. 11/15 systems yield lower raw energy than the NOON-MP2 baseline while preserving the chemical manifold. The framework is not optimised solely for energy; when energy improvement occurs, it does so together with, not instead of, chemical meaningfulness.
- Multi-basin exploration is a feature, not a defect. For Pyrrole, Imidazole, and Thioformaldehyde, seeds fall into distinct CASSCF basins — all chemically valid, some significantly lower in energy than NOON-MP2. This demonstrates that combinatorial search accesses regions of active-space landscape that local orbital rotations cannot reach.
- 15 systems tested — mostly small-to-medium organic molecules (4–16 electrons in active space).
- Transition-metal coverage remains preliminary (1 system, Cr₂, under reduced basis STO-3G). A comprehensive transition-metal benchmark would require multiple metals, larger basis sets, and more geometries.
- Aromatic systems require NOON-MP2 injection at the current compute budget. No-injection performance at larger population sizes is untested.
- All systems tested are ground-state (S₀) or lowest few states. Excited-state manifold consistency under SA-CASSCF requires separate evaluation.
- These results demonstrate broad practical robustness across the tested chemical families, not formal universality. Extrapolation to untested classes (large conjugated systems, actinides, open-shell ground states) should be made cautiously.